- News & information
- About
- History
- George V. Voinovich
- George V. Voinovich Collection
- Calendar
- How to Find Us
- News
- Archives
- Photojournalism Fellowship Project
- Photo Essays
- Current Fellow
- Previous Fellows
- Reports and Publications
- Archives
- Students
- Prospective
- Center for Entrepreneurship
- Environmental Studies
- HTC/Voinovich School Scholars
- Master of Public Administration
- Current
- HTC/Voinovich School Scholars
- Center for Entrepreneurship
- Environmental Studies
- Master of Public Administration
- Alumni
- Contact
- School Leadership
- Strategic Partners Alliance
- Ohio University Public Affairs Advisory Committee
- Ohio University Public Affairs Advisory Committee
- Faculty and Fellows
- Faculty
- Visiting Professors
- Voinovich Fellows
- Professional Staff

Review: Light
Review: Light

- Light shows wave properties
- Diffraction: waves spread out after they pass through an aperture (small opening)
- Refraction: waves will `bend' when going through certain materials
- What is light?
- An electric `field' surrounds all charged objects
- An oscillating charge will produce as oscillating electro-magnetic field The oscillating electro-magnetic field propagates as transverse wave.
- Electro-magnetic waves of a certain frequency (wavelength) are perceived as `light'.
- We perceive different frequencies of light as different 'colors'
- All hot objects emit light.
- A black-body is a body that absorbs all radiation that falls on it.
- When a black body is heated, it starts to glow and emits a characteristic black-body spectrum.
- More energy is emitted from hot objects than from cool ones. The peak emission occurs at higher frequencies for hot objects, at lower frequencies for cooler objects.
- An electron, such as those in the atoms in your eye, will be pulled along with the oscillating electric field. This is how we `see' light
- The electrons in an atom or molecule can absorb or emit photons (light quanta of a certain energy) and thus produce a discrete spectrum.
- Emission spectrum: Atoms or molecules emit photons (light quanta) of a certain discrete energy when the electrons jump from a higher level to a lower level. These photons have an energy characteristic for the atom or molecules, thus show distinct colors in the visible spectrum.
- Absorption spectrum: Atoms or molecules absorb photons of a certain energy, thus there are black lines (missing frequencies) in the light spectrum.
- Light wavelengths
- The wavelength is related to the frequency be
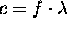
- The speed of light in the vacuum is c = 3 * 10 8 m/s .
- Very long wavelengths of light are called radio waves
- Long wavelengths of light are called infrared.
- Visible light has wavelengths of 400 to 700 nanometers (a nanometer is one billionth of a meter, or 10 -9 meter).
- Short wavelengths of light are called ultraviolet
- The wavelength is related to the frequency be
- Light Sources
- Incandescent light: produce light through being heated, e.g. light bulbs, candles.
- Fluorescence: The process of absorbing ultraviolet light and emitting visible light. Excited atoms in fluorescent materials drop to their normal level in about 10 -8 s.
- Phosphorescent materials continue to glow long after the illumination has been removed. The atoms remain in an excited metastable state for periods ranging from a few seconds to several hours.
- Triboluminescence: The process of producing light by mechanical forces exerted on an object (e.g. crushing a wintergreen candy).
- Photometry
- Intensity is power per unit area and is measured in cd [candles].
- Luminance: intensity per unit area [candles/m 2 ].
- The intensity falls of inversely proportional to the square of the distance from the light source.
 Ch. Elster
Ch. ElsterAug 26 14:27:03 EDT 2019
Contact Information:
(740) 593–9381 | Building 21, The Ridges
Ohio University Contact Information:
Ohio University | Athens OH 45701 | 740.593.1000 ADA Compliance | © 2018 Ohio University . All rights reserved.
